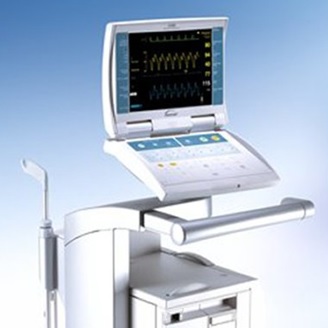
CS100i, CS100, and CS300 Intra-Aortic Balloon Pumps
Datascope Corp. and MAQUET expanded a recall issued earlier this year to include 12,319 units of the CS100i, CS100, and CS300 Intra-Aortic Balloon Pumps that were manufactured from July 1, 2003 through June 16, 2017.
The FDA warns that “device failure may result in immediate and serious adverse health consequences, including death” for patients who require circulatory support to survive.
Electrical failure of the pump is associated with 1 patient death, according to the manufacturer:
The device failed to pump due to an electrical test failure … which has been associated to a patient death due to the failure of the device to initiate therapy.”
The problem with the devices is a “false blood-detection alarm and ingress of fluid” into the Intra-Aortic Balloon Pump, according to the FDA. The recall is designated as Class 1 by the FDA, which is the highest level of safety recall.
In June 2013, Datascope Corp./MAQUET implemented a design change to prevent the electrical failure that prevents the balloon from inflating or deflating. The problem is that not all devices manufactured before June 13, 2013 were serviced or upgraded.
For more information, consumers can contact Datascope Corp./MAQUET’s Customer Service Department at 1-(888)-627-8383 and press “2” (Monday through Friday from 8:00 a.m. to 6:00 p.m. EDT).
