The lawsuit was filed by Aleasia J. P., a woman who was implanted with the C.R. Bard Meridian® Vena Cava Filter on October 12, 2012 at a hospital in Virginia.
C.R. Bard no longer manufactures or sells the Meridian in the United States. The company is facing over 1,000 lawsuits involving the Meridian and other IVC filters. Lawyers accuse the company of negligence for selling defective medical devices and downplaying serious safety risks.
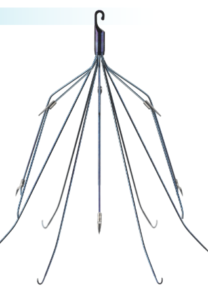
C.R. Bard Meridian® IVC Filter
Meridian is a temporary filter that consists of 12 needle-like legs arranged in a cone-shape around a central hook. It is implanted in a major blood vessel called the inferior vena cava (IVC) to catch blood clots and prevent pulmonary embolisms
Meridian was approved by the FDA in 2011 with a 510(k) application, which means it did not go through clinical trials because it was “equivalent” to several other IVC filters made by C.R. Bard. Unfortunately, these other filters have been linked to serious risks.
Meridian is highly-similar to the Recovery, which was introduced in 2003 and withdrawn in the market in 2005. In 2012, a study of 363 patients estimated that 40% would fracture within 5.5 years.
Meridian is also very similar to the G2, which C.R. Bard introduced in 2005 to replace the Recovery. In June 2014, a study of 829 patients implanted with the G2 estimated that 38% would fracture within 5 years, with the risk increasing over time.
Fracture is a very serious risk associated with IVC filters, especially temporary filters that are not removed within 29-54 days of implantation. The broken needle-like legs can travel in the bloodstream, get trapped in the heart or lungs, and cause severe irregular heart rhythm or sudden death.
The lawsuit was filed on November 8, 2016 in the U.S. District Court for the District of Arizona — Case No. 2:16-cv-03871.
It will be centralized in Multi-District Litigation (MDL No. 2641)— In Re: Bard IVC Filters Products Liability Litigation.
The plaintiff is represented by Ben C. Martin of The Law Offices of Ben C. Martin in Dallas, Texas.
 Editor’s note: For more information about IVC Filter lawsuits and your legal rights, please contact The Law Offices of Ben C. Martin.
Editor’s note: For more information about IVC Filter lawsuits and your legal rights, please contact The Law Offices of Ben C. Martin.
