The lawsuit was filed by the family of Robert B., a deceased man who was injured by the Meridian® Inferior Vena Cava Filter (“IVC Filter”) manufactured by C.R. Bard and Bard Peripheral Vascular, Inc.
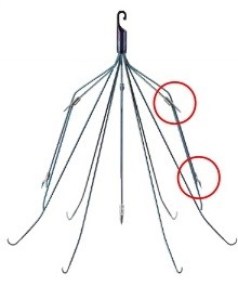
C.R. Bard Meridian® IVC Filter
The IVC filter was implanted in his body at a hospital in Illinois on December 20, 2013 for the purpose of preventing a pulmonary embolism, or blood clots in the lungs.
Meridian is a temporary IVC filter with a hook at the tip to facilitate retrieval when it is no longer needed. It is a 5th-generation Bard IVC filter, but the design is nearly identical to previous generations.
Like these previous generations, Meridian was approved without clinical trials under the FDA’s 510(k) process. It was also pulled off the market without a recall a few years after it was approved in 2011.
The only major design changes between the Meridian and previous generations is that it has additional barbs near the hook that help anchor it inside the inferior vena cava to reduce tilting rates.
Unfortunately, these design changes did not address the most serious problems associated with Bard’s 1st-generation Recovery IVC filter (withdrawn in 2005) and the 2nd-generation G2 IVC filter (withdrawn in 2010). Both of these older filters are estimated to fracture in around 40% of patients within 5 years of implantation, according to recent studies.
For example, in one recent case report, a 74 year-old woman was diagnosed with a fractured Meridian IVC filter within 8-12 months after it was implanted in her body. The researchers also noted 38 reports of Meridian IVC filter fractures in 2013 alone.
Fracture is an extremely serious complication that can cause sudden death. Broken parts of the filter will travel in the bloodstream until they get stuck somewhere, often in the heart, lungs, or other major organs.
Another frequently deadly long-term complication is blood clots. The longer a filter is implanted, the higher the risk of developing Deep Vein Thrombosis (DVT). Blood clots might also form on the exposed metal legs of the IVC filter, a common problem with metal implants. If these blood clots break loose, they can cause a pulmonary embolism.
The lawsuit was filed on April 17, 2017 in the U.S. District Court for the District of Arizona — Case No. 2:17-cv-01134.
Approximately 1,700 other IVC filter lawsuits have been filed against C.R. Bard in a federal Multi-District Litigation (MDL No. 2641) — In Re: Bard IVC Filters Products Liability Litigation.
The plaintiff is represented by Ben C. Martin of The Law Offices of Ben C. Martin in Dallas, Texas. He serves on the plaintiffs’ steering committee of the Bard IVC Filter MDL.
 Editor’s note: For more information about IVC Filter lawsuits and your legal rights, please contact The Law Offices of Ben C. Martin.
Editor’s note: For more information about IVC Filter lawsuits and your legal rights, please contact The Law Offices of Ben C. Martin.

