Medtronic Strata® adjustable pressure valves are surgically implanted in the skull of people with hydrocephalus, or “water on the brain,” a congenital condition that can cause brain damage if left untreated.
The valves are part of a shunt system that drains fluid in the skull down through a tube to the abdomen, where the fluid is re-absorbed.
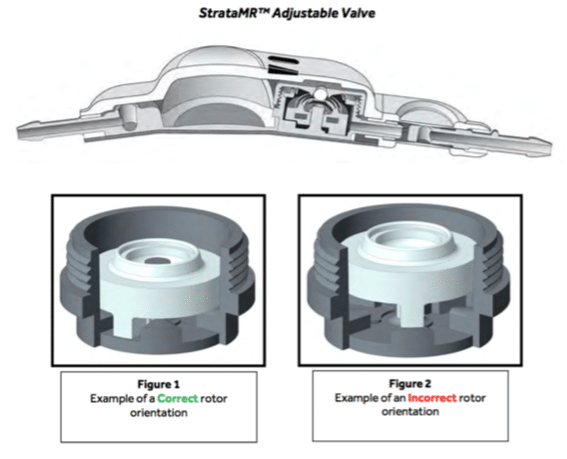
Unlike fixed-pressure shunt valves that must be carefully calibrated and adjusted surgically, Strata MR valves are adjusted non-surgically using magnets. The doctor simply places a strong magnet on the back of the patient’s skull and turns the dial to calibrate the pressure.
The problem is that drainage problems can occur if the mechanism is adjusted to a position that causes too much flow-resistance. This can potentially lead to coma and death as pressure increases in the skull.
There has been one reported patient death, but the cause of death has not been confirmed to be related to the malfunctioning valve.
Medtronic initiated the recall on February 22, 2017 with an Urgent Field Safety Notice to surgeons. On April 8, the FDA warned:
Under-drainage of cerebrospinal fluid may result in the following adverse health consequences: headaches, nausea, vomiting and lethargy. If left untreated, under-drainage can potentially lead to coma and death.”
The product complaint-rate related to this issue was 2.75% of the total number of units that have been distributed (approximately 72 reports).
The recall involves about 2,622 StrataMR valves and shunts with model numbers 42955, 42965, 45905, 46955, 46960, 46965, and 46970 manufactured from October 27, 2015 to November 11, 2016.
Patients with the valves who experience headaches, nausea, vomiting, lethargy, or symptoms of under-drainage of cerebrospinal fluid should consult the physician who implanted the StrataMR valve.

Use Aesculap’s progav2. Much better and easy to adjust.
What if mine was put in September 2015 and I’m having problems with shunt. Unable to reset it to appropriate pressure.